DOCUMENT CONTROL
SOPs/STPs, BMR/BPR, Specifications, Change History Management. Foundation of GMP compliance.
2
VENDOR QUALIFICATION
Vendor Evaluation, Audit & Approval, Material Approval Status. Ensures supply chain quality.
3
RAW MATERIAL CONTROL
Review COA, Sampling Authorization, Release/Reject Decision. First point of quality gatekeeping.
4
IN-PROCESS QA
Line Clearance, Process Monitoring, In-Process Checks. Real-time quality oversight during manufacturing.
5
DEVIATION MANAGEMENT
Deviation Identification, Root Cause Analysis, Impact Assessment. Handling unexpected events.
6
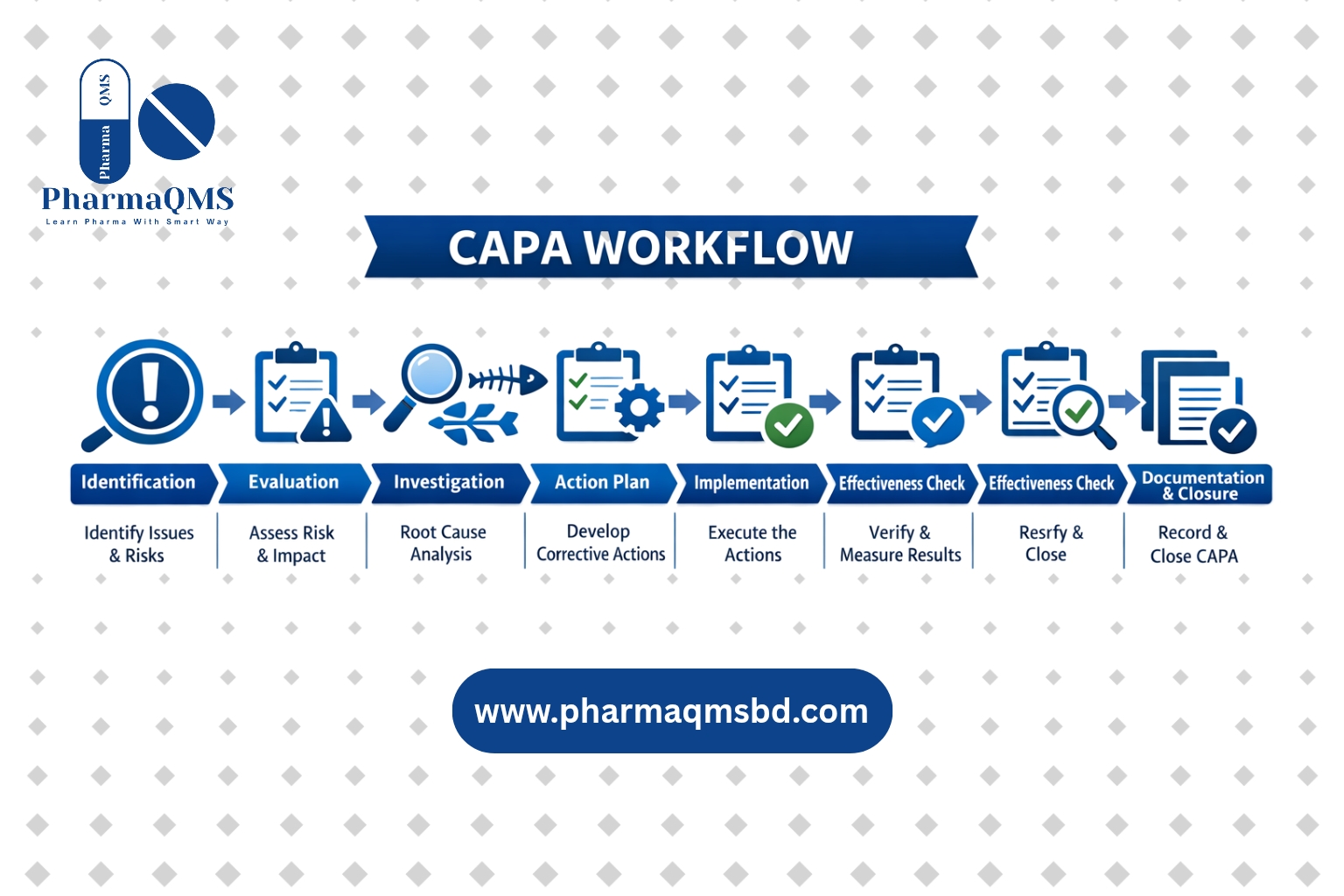
CAPA
Corrective & Preventive Actions, Effectiveness Check. Systematic problem-solving and prevention.
7
CHANGE CONTROL
Change Proposal, Risk Assessment, Approval & Implementation. Managed evolution of systems.
8
VALIDATION & QUALIFICATION
Process Validation, Cleaning Validation, Equipment Qualification. Proving systems work consistently.
9
AUDIT & COMPLIANCE
Internal Audit, Regulatory Audit, GMP Compliance. Verification and adherence to standards.
10