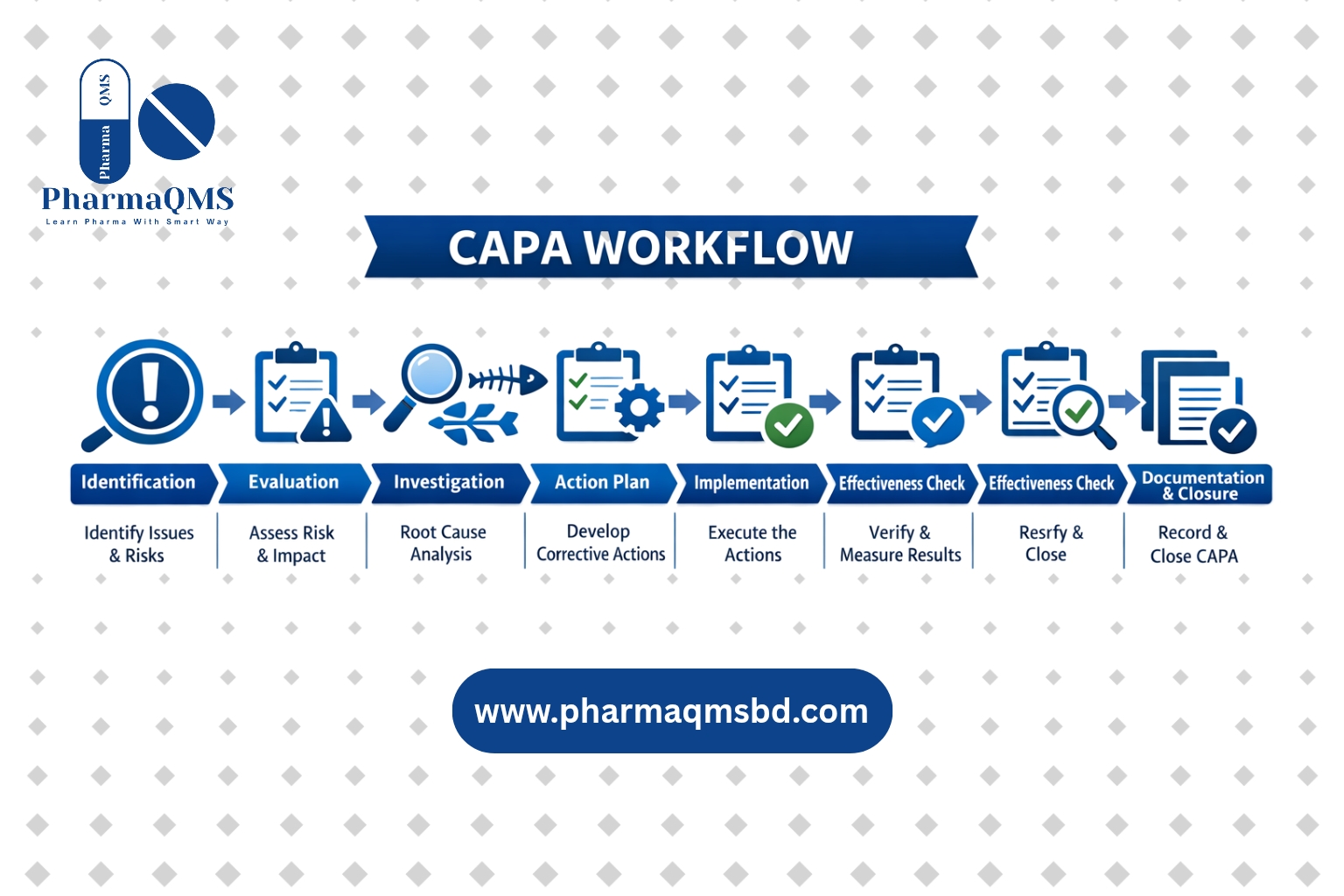
What is change control
A Process which ensures that changes to procedures, materials, methods, equipment, and software are properly documented, approved, validated and traceable.
CHANGE CONTROL PROCEDURE:
DEFINATION: Change Control: A formal system by which qualified representative of appropriate disciplines review proposed or actual changes that might affect the validated status of facility, systems, equipments or processes.
Temporary Change: A change (departure from any established procedure/ system/ process) initiated for the evaluation of proposed procedure/system/process, which has been taken with prior approval to achieve the desired output, allowed for one time change and limited to a particular batch. For example change in batch size, manufacturing equipment, etc.
Permanent change/Critical Change: A change initiated based upon scientific rational or historical GMP data or data generated through temporary changes.
Major Change: Changes, proposed for improvements to process, materials, product and procedures which may have impact upon the identity, quality, purity, strength, stability, safety and efficacy or physical characteristic of the product. Notification to agency required.
Minor Change: Changes, which does not have impact on the quality attributes like identity, quality, purity, strength, stability, safety, efficacy or physical characteristic of the product.
The change control approval or rejection process shall require to be completed within 30 working days from the date of initiation of the change control.
Change control preferably closed within 90 working days after Head –QA approval.
If change control is not closed within specified timeline, initiator shall raise “Period Extension Request” as per SOP.
Initiating department Head shall review the extension request and write justification for delay with impact assessment. QA shall assess the impact of delay in action completion and approve / reject the Period extension request. Period extension shall be allowed for two times only. After this new change control shall be initiated.
CLASSIFICATION OF TYPICAL CHANGES
- Change in systems (Major)
- Change in manufacturing formula/process / New Products (Critical & Major)
- Change in expiry (related to stability) (Critical & Major)
- Change in critical Raw Material/solvent (Critical & Major)
- Change in specifications and test method (Critical & Major)
- Change in SOP for addition / deletion (Major & Minor)
- Change in equipment (Major)
- Modification in critical equipment (Major)
- Modification / Up gradation in facility (Major)
- Change in stability program (Critical & Major)
- Change in key raw material source or supplier (Major)
- Change in storage conditions (Major)
- Change in primary packing material (Major)
- Change in secondary packing material (Minor)
- Change in packing style (Minor)
- Change in printed text on label (Major)
- Change in manufacturing location/site (Critical & Major)
- Change in manufacturing Batch Size (Critical & Major)
- Change in packing batch size (Minor)
- Change in control systems i.e. computers, Data Collection (Major)
- Formats and internal labels (Minor)
- Deletion of a product (Minor)
Note: The list can be elaborated based on practical changes occurring at the locations.
Product Change : Change in key RM/Solvent, BOM, Process Parameters, In-process control, pack style, packing material, introduction of New Product etc
Engineering Change: Change in Facility design, equipment type, Maintenance parameters, utilities.
System Change: Change in software/firmware or its configuration etc. Documentation Change: Change in SOP, STP, Document control procedures etc.