Pharmaceuticals Water System (WFI and WTP) - Short Note
lasima Anjum Naima, Lecturer, Dept. of Pharmacy
4 Sources of Water
- Underground Water
- Rainwater
- Seawater
Types of Pharmaceutical Water
- Non-Potable Water - Not suitable for drinking or pharmaceutical use.
- Potable (Drinkable) Water - Safe for human consumption.
- USP Purified Water - Purified for pharmaceutical preparations.
- USP Water for Injection (WFI) - Sterile and pyrogen-free water for injection purposes.
- USP Sterile Water for Injection - Sterile water intended for injection.
- USP Sterile Water for Inhalation - Sterile water used in inhalation therapy.
- USP Bacteriostatic Water for Injection - Sterile water containing antimicrobial agents to inhibit bacterial growth.
- USP Sterile Water for Irrigation - Sterile water used for surgical or medical irrigation.
Water is a vital resource in industrial settings, especially in sectors like pharmaceuticals, food & beverage, and power generation. Two key systems are used to ensure water quality and environmental compliance: the Water Treatment Plant (WTP) and the Effluent Treatment Plant (ETP). While WTPs purify raw water for use, ETPs treat wastewater before its disposal or reuse.
Principles for Pharmaceutical Water Systems
1. Design and Maintenance
Pharmaceutical water production, storage, and distribution systems must be designed, installed, commissioned, qualified, and maintained to ensure reliable production of water with the required quality. The water production process must be validated to confirm that the system operates within its designed capacity and consistently meets quality specifications.
2. Monitoring
Water sources and treated water should be regularly monitored for:
- Chemical contamination
- Microbiological contamination
- Endotoxins, when applicable
The performance of purification, storage, and distribution systems should also be routinely assessed to ensure proper functioning.
3. Documentation and Trend Analysis
All monitoring results, trend analyses, and any corrective actions taken should be properly recorded and maintained for quality assurance and regulatory compliance.
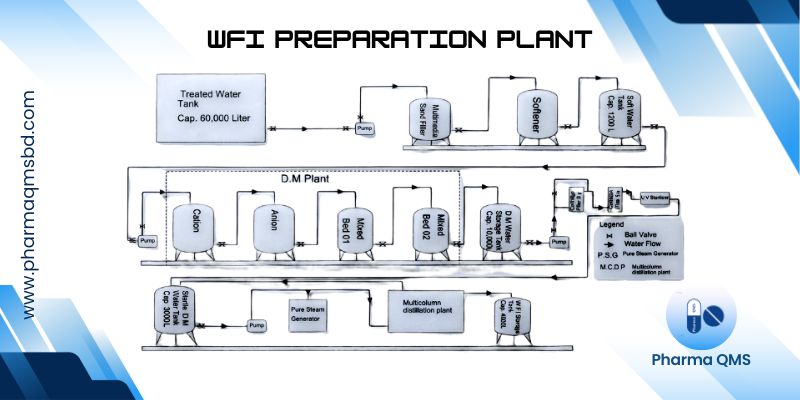
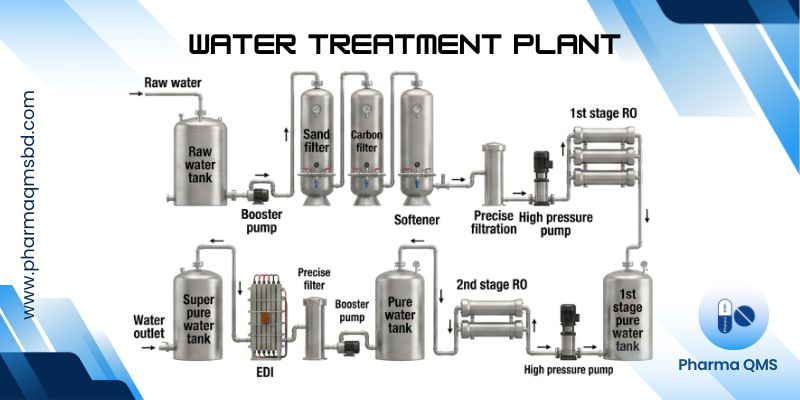
Water Treatment Plant (WTP)
Water treatment in the pharmaceutical sector refers to the series of processes designed to remove impurities, contaminants, and microorganisms from water to meet specific quality standards suitable for various pharmaceutical applications.
Operation of a Water Treatment Plant (WTP)
1. Water Intake/Source
Raw water is collected from sources such as groundwater, surface water, or rainwater. Pre-screening may remove large debris and suspended solids.
2. Pre-Treatment
Sedimentation / Coagulation & Flocculation: Chemicals may be added to aggregate fine particles into larger flocs for easier removal. Pre-Filtration: Removes sand, silt, and other suspended solids.
3. Primary Purification
Filtration: Through multimedia or cartridge filters to remove fine particulates.
Ion Exchange / Deionization: Removes dissolved ions to reduce water hardness and total dissolved solids.
Reverse Osmosis (RO): High-pressure filtration to remove salts, organic compounds, and microorganisms.
Distillation (optional): Produces highly purified water by boiling and condensing.
4. Disinfection
UV Treatment: Kills or inactivates microorganisms.
Chemical Disinfection: Sometimes used for potable water but less common in WFI systems.
5. Storage
Purified water is stored in dedicated tanks made of materials that prevent contamination. Tanks are designed to avoid stagnation and microbial growth.
6. Distribution
Water is circulated through a looped distribution system to maintain quality. System includes pumps, pipes, and recirculation loops.
7. Monitoring and Quality Control
Chemical, microbial, and endotoxin testing are performed regularly. Trend analysis ensures the system operates within validated specifications.
Water Treatment Methods/techniques in Pharmaceuticals
- Ion Exchange / Deionization (DI) – Removes dissolved ions to produce purified water.
- Electrodeionization (EDI) – Continuous removal of ions using electricity and ion-exchange resins.
- Filtration – Physical removal of particulate matter and microorganisms.
- Ultraviolet (UV) Treatment – Disinfection by destroying microbial DNA.
- Distillation – Boiling and condensing water to remove impurities and contaminants.
- Reverse Osmosis (RO) – Pressure-driven filtration to remove dissolved salts, organic matter, and microorganisms.
Demineralization (DM Water)
- Removal of dissolved mineral salts from water by ion exchange, distillation, membrane filtration, or electrodialysis.
- Also called Deionized (DI) water.
Deionization
A physical process using specially manufactured ion exchange resins.
Resins exchange ions in water with H+ and OH− ions, which then recombine to form pure H2O.
Since most water impurities are salts, this process produces high-purity water comparable to distilled water, but it is faster and avoids scale buildup.
Ions Removed
Cations: Ca2+, Mg2+, Na+, K+
Anions: Cl−, HCO3−, NO3−, CO32−
Ion Exchange Resins
1. Cation Exchange Resins
Release H+ ions (or other positive ions) in exchange for impurity cations.
Example: Ca2+ in water gets replaced by 2H+.
2. Anion Exchange Resins
Release OH− ions (or other negative ions) in exchange for impurity anions.
Example: Cl− in water gets replaced by OH−.
3. Types of Resin Applications:
- Cation Exchange Alone (Single Bed): Used for water softening by replacing Ca2+ and Mg2+.
- Anion Exchange Alone (Single Bed): Removes nitrates, sulfates, and organics.
- Mixed-Bed Exchange (Cation + Anion Resins together): Produces the highest purity water.
Electrodeionization (EDI)
- Definition: A continuous water purification process combining ion-exchange resins with an electric field to remove ions.
- Purity: Produces high-purity water with specific resistance up to 10 MΩ·cm or more.
- No Chemicals: Unlike conventional ion exchange, it requires no chemical regeneration—electric current regenerates the resins.
- Cost-Effective: Lower running cost compared to traditional ion-exchange methods.
- Easy Operation: No chemical handling makes operation and maintenance simple.
1. Sand Filter
Removes large particles, suspended solids, and turbidity. Works as a mechanical barrier for coarse impurities.
2. Carbon Filter
Uses activated carbon bed to remove organic compounds, chlorine, and odors. Works by chemical adsorption of contaminants.
3. Pre-Filter
Initial stage filter to remove larger particles before fine filtration. Protects delicate downstream filters (e.g., RO membranes).
4. Membrane Filter
Pore size: 0.22–0.45 μm, thickness ~150 μm. Removes bacteria and fine particles. Commonly used for sterile filtration in pharmaceutical processes.
5. Ultra Filter (UF)
Pore size: ~0.01 μm. Removes microorganisms (bacteria, protozoa), but viruses may still pass. Microfiltration (~0.1 μm) removes larger microbes but less effective against viruses.
6. UV Filter (Ultraviolet Treatment)
- 185 nm: Breaks down Total Organic Carbon (TOC) into CO2 and H2O.
- 254 nm: Destroys microbial DNA, preventing reproduction (disinfection).
Reverse Osmosis (RO)
1. Principle:
Uses a semipermeable membrane and high pressure to force water through while rejecting dissolved salts, organics, and microorganisms.
2. Process:
Water flows under pressure → passes through RO membrane → pure water (permeate) is collected, while concentrated impurities (reject/brine) are discarded.
3. Features & Advantages:
- Removes 90–99% of dissolved salts.
- Effective against bacteria, pyrogens, and particulates.
- Energy-efficient compared to distillation.
- Requires pretreatment (sand/carbon filters) to protect membranes.
Distillation
1. Principle:
Involves boiling water to produce steam, then condensing the steam to obtain pure water.
2. Process:
Raw water → heated to vaporize → steam rises → condensed into liquid form → pure distilled water.
3. Features & Advantages:
- Removes salts, heavy metals, organic impurities, and microbes.
- Produces very high-purity water, often used for Water for Injection (WFI).
- More reliable for pyrogen removal compared to other methods.
4. Limitations:
- Energy-intensive (high operational cost).
- Requires cooling and large equipment space.
Applications of WTP
- Pharmaceutical Industry: Provides purified water for manufacturing processes, cleaning, and laboratory applications.
- Food & Beverage: Ensures water is safe and meets quality standards for processing.
- Hospitals and Laboratories: Supplies sterile and safe water for medical use.
- Power Plants: Prevents scaling and corrosion in boilers and cooling towers.
Effluent Treatment Plant (ETP)
An Effluent Treatment Plant (ETP) is essential in pharmaceutical manufacturing to treat and safely dispose of wastewater containing active pharmaceutical ingredients (APIs), heavy metals, toxic chemicals, organic matter, and microbial contaminants. Proper effluent treatment ensures compliance with environmental regulations (e.g., EPA, CPCB, WHO) and prevents water pollution.
Operation of an Effluent Treatment Plant (ETP)
1. Preliminary Treatment
Removal of large solids, grit, and floating matter by screening and grit chambers. Equalization tanks balance pH and flow rate of incoming effluent.
2. Primary Treatment
Sedimentation/Clarification: Heavier solids settle at the bottom as sludge. Oil and grease are skimmed off the surface.
3. Secondary (Biological) Treatment
Uses microorganisms (aerobic or anaerobic) to degrade dissolved organic matter. Common methods: Activated Sludge Process, Trickling Filters, Anaerobic Digesters. Produces treated water and biological sludge.
4. Tertiary Treatment (Advanced Treatment)
Further purification for removal of fine solids, nutrients, and pathogens. Includes filtration, activated carbon, UV treatment, chlorination, or RO. Ensures water meets discharge or reuse standards.
5. Sludge Treatment & Disposal
Sludge from primary and secondary stages is thickened, digested, and dewatered. Safely disposed of, incinerated, or used as fertilizer (if non-toxic).
6. Final Discharge
Treated effluent is discharged into surface water bodies, sewer systems, or reused in industries depending on quality standards.
1. Physical Techniques
- Screening: Removes large floating solids, plastics, and debris.
- Sedimentation: Heavier particles settle at the bottom as sludge.
- Filtration: Removes suspended solids and fine particles.
- Equalization: Balances variations in effluent flow and pH.
2. Chemical Techniques
- Coagulation & Flocculation: Addition of chemicals (alum, ferric salts, polyelectrolytes) to aggregate fine particles for easy removal.
- Neutralization: Adjusting pH using lime, caustic soda, or acids.
- Oxidation/Reduction: Chemical oxidants (chlorine, ozone, hydrogen peroxide) degrade toxic pollutants.
- Precipitation: Converts dissolved contaminants into insoluble forms for removal.
3. Biological Techniques
- Aerobic Treatment: Uses oxygen-dependent microbes (e.g., activated sludge, trickling filters) to degrade organic matter.
- Anaerobic Treatment: Microbes degrade organics without oxygen (e.g., anaerobic digesters, UASB reactors).
- Stabilization Ponds/Lagoons: Natural, low-cost biological treatment using sunlight, algae, and microbes.
4. Advanced / Tertiary Techniques
- Membrane Processes: Reverse Osmosis, Ultrafiltration, Nanofiltration.
- Adsorption: Activated carbon removes color, odor, and organics.
- Ion Exchange: Removes dissolved salts and specific contaminants.
- UV / Ozone Treatment: Disinfection and removal of pathogens and toxic organics.
API manufacturing wastewater treatment is a type of effluent treatment, because:
- It deals with industrial wastewater from pharmaceutical processes (chemical synthesis, fermentation, etc.).
- The wastewater is complex, containing high BOD, COD, TSS, toxic chemicals, solvents, and variable pH.
- Treatment requires a specialized Effluent Treatment Plant (ETP), combining physical, chemical, biological, and advanced processes.
Sources of Wastewater in API Manufacturing:
1. Chemical Synthesis / Semi-Synthesis Processes
Process: Involves multiple reaction steps → isolation of products by liquid-liquid extraction, leaching, crystallization, and filtration.
Type of Effluents Generated:
High BOD & COD: Because of organic solvents, reagents, and unreacted chemicals.
High TSS (Total Suspended Solids): From filtration residues and crystallization byproducts.
Wide pH range (1-11): Due to use of acids, bases, and catalysts.
2. Fermentation Process
Process: Biochemical reaction using microorganisms (yeast, bacillus, etc.). Steps → inoculum preparation, fermentation, product recovery.
Type of Effluents Generated:
Organic-rich wastewater (high BOD/COD): From unutilized nutrients, microbial biomass, residual sugars.
Suspended Solids: From cell debris and filtration steps.
Chemicals/Disinfectants: Solvents, phenolic compounds, salts, and ion exchange resins.
Treatment Techniques in ETP for API Wastewater / Treatment of Pharmaceutical Waste Water:
- Physical Treatment: Screening, sedimentation, and filtration to remove solids and biomass.
- Chemical Treatment: Neutralization: Adjust pH. Coagulation/Flocculation: Remove suspended solids. Oxidation/Reduction: Degrade toxic organics and solvents.
- Biological Treatment: Aerobic processes: Activated sludge, trickling filters for biodegradable organics. Anaerobic processes: Digest organic-rich streams and reduce sludge volume.
- Advanced/Tertiary Treatment: Adsorption (activated carbon) for organics and color removal. Membrane processes (RO, UF) for salts and fine impurities. UV or ozone for disinfection and TOC reduction.
Sludge Handling
Sludge from primary, secondary, and tertiary treatments is thickened, dewatered, and safely disposed or recycled.
Applications of ETP
- Pharmaceutical & Chemical Plants: Treats effluents containing hazardous chemicals and active ingredients.
- Textile and Dyeing Industries: Removes dyes and chemical residues from wastewater.
- Food & Beverage: Processes organic waste and other contaminants from production facilities.
- Hospitals and Laboratories: Treats potentially infectious or hazardous wastewater.
Key Differences Between WTP and ETP
| Feature | Water Treatment Plant (WTP) | Effluent Treatment Plant (ETP) |
|---|---|---|
| Purpose | Purifies raw water for industrial use | Treats wastewater before discharge or reuse |
| Main Processes | Screening, coagulation, sedimentation, filtration, disinfection, RO | Pre-treatment, primary (chemical/setting), secondary (biological), tertiary (advanced filtration/disinfection) |
| Output | High-quality, usable water | Treated, environmentally safe water |


